1832
Views & Citations832
Likes & Shares
Treatment of the ketones 1, 5, 7 with Grignard reagents yielded
the products 4, 6, 8, respectively. The
ester 9 with Grignard reagent
followed by alkaline hydrolysis afforded lactones 10 and 11, respectively.
The lactones 14 and 17 on being treated with Grignard
reagent furnished the diketone 15
and the chloroketone 18 respectively.
The epoxide 19 with Grignard reagent
produced diol 21 whereas the epoxide
22 suffered ring contraction
yielding the aldehyde 23.
Keywords: Methylmagnesium bromide, Methylmagnesium
iodide, Phenylmagnesium chloride, t-Butyl magnesium bromide, Epoxides, Lactones
The reactions of Grignard reagents with carbonyl compounds are well documented [1,2]. The resulting products have been utilized for the synthesis of many natural products and many bioactive organic compounds. Though there are many Grignard reagents in this this micro review only the reaction of methyl magnesium bromide (MeMgBr) and methyl magnesium iodide (MeMgI) with carbonyl compounds have been discussed. The reaction of phenyl magnesium bromide (PhMgBr) and t-butyl magnesum bromide (t-BuMgBr) have been discussed briefly. The Grignard reagents are represented as R-Mg-X, where R is alkyl, alkenyl, alknyl aryl or t-butyl group and X is Cl, Br or I. For an alkyl halide, the ease of formation of Grignard reagent is of the RI>RBr>RCl. The reaction is performed under anhydrous conditions. In case the reaction is sluggish, a small amount of iodine is added to start the reaction. The workup of the Grignard reagents (MeMgI, MeMgI) is usually done by adding an aqueous solution of HCl or H2SO4. When the products cannot tolerate acids, however a large volume of saturated aqueous ammonium chloride solution is added to dissolve all magnesium salts. The formation of the product depends on: (a) the nature of the functional groups present in the organic compound; (b) the kind of Grignard reagents used; and, (c) reaction condition. It has been observed that the Grignard reagents with some ketones, esters, epoxides, lactones have afforded unexpected product instead of expected normal product.
With ketone
(i) It is known that ketone with the Grignard
reagents yield alcohol which on dehydration affords alkene. An interesting
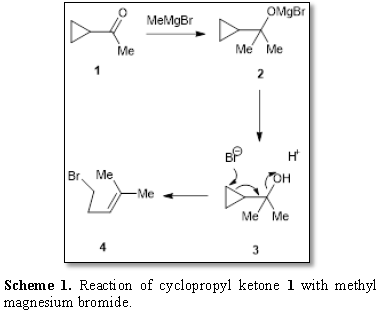
observation was recorded as result of the reaction of cyclopropyl ketone 1 with methyl magnesium bromide [3].
The resulting product 2 was treated
with aqueous hydrogen bromide. The expected alcohol 3 was not obtained because the ring cleavage occurred by Br-
anion affording 4 (Scheme 1).
Scheme 1. Reaction of cyclopropyl ketone 1 with methyl magnesium bromide.
The reaction is an example of the ready
rearrangement of cyclopropyl-carbinyl derivative to allyl carbonyl product [4].
The halide can react with Grignard reagent and used to repeat the process. In
this way a chain of isopropenoids units can be built
up. (ii) The
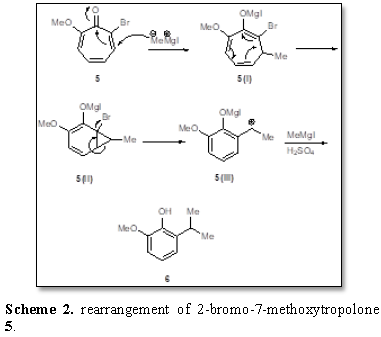
rearrangement of 2-bromo-7-methoxytropolone 5 was observed when treated with methyl magnesium iodide and diluted
sulfuric acid [5]. The normal product was not obtained. The resulting product
was identified as 2-hydroxy-3-isopropylanisole 6 (Scheme 2). The intermediate 5(i)
is formed from a nucleophilic attack by the methylmagnesium iodide on the
carbonyl carbon of the tropolone ring and this was followed by ring contraction
yielding the intermediate 5(ii). The
elimination of the halogen atom yields the anion 5(iii) which reacts with another molecule of methylmagnesium iodide
to afford the isopropylanisole 6. The
formation of 6 is an interesting
example [6] of the rearrangement of the troponoid occurring by means of
nucleophilic reagents.
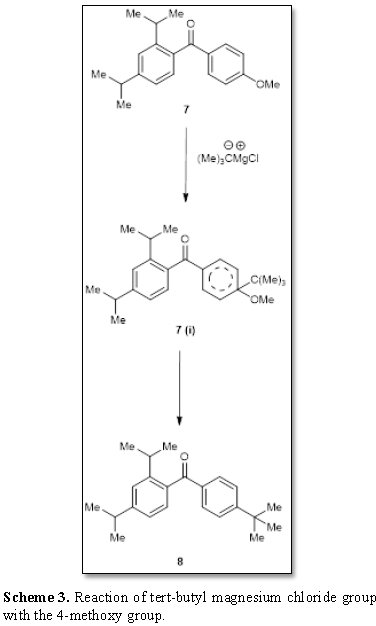
iii) The reaction of the ketone [7] 7 with t-butyl magnesium chloride afforded an interesting product which was assigned to the structure 8. The formation of 8 shows that due to steric hindrance the bulky Grignard reagent failed to attack the carbonyl group. The formation of 8 has been explained in Scheme 3. The Grignard reagent tert-butyl magnesium chloride group attacks the 4-methoxy group and forms the intermediate 7 (i) which is finally converted (as shown in Scheme 3) into the compound 8.
Scheme 2. rearrangement of 2-bromo-7-methoxytropolone 5.
iii) The reaction of the ketone [7] 7 with t-butyl magnesium chloride afforded an interesting product which was assigned to the structure 8. The formation of 8 shows that due to steric hindrance the bulky Grignard reagent failed to attack the carbonyl group. The formation of 8 has been explained in Scheme 3. The Grignard reagent tert-butyl magnesium chloride group attacks the 4-methoxy group and forms the intermediate 7 (i) which is finally converted (as shown in Scheme 3) into the compound 8.
With ester
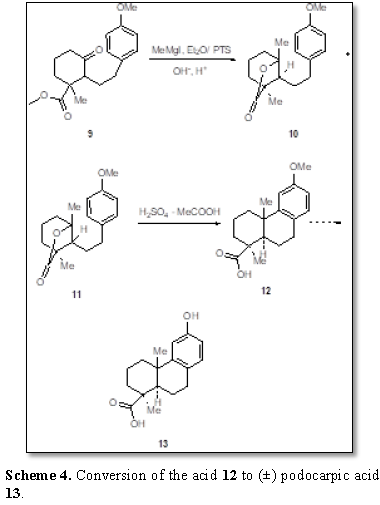
The reaction of the Grignard reagent with the ketoester 9 is very interesting [8]. The ketoester 9 on being treated with excess methylmagnesium iodide followed by
heating with p-toluene sulfonic acid yielded a dense material which on partial
alkaline hydrolysis and acidification with hydrochloric acid yielded a mixture
of lactones 10 and 11. The mixture of lactones on
cyclization with sulfuric acid and acetic acid afforded 12-methoxypodocarpic
acid 12. The conversion of the acid 12 to (±)
podocarpic acid 13 has already been
reported [9] (Scheme 4).
With lactones
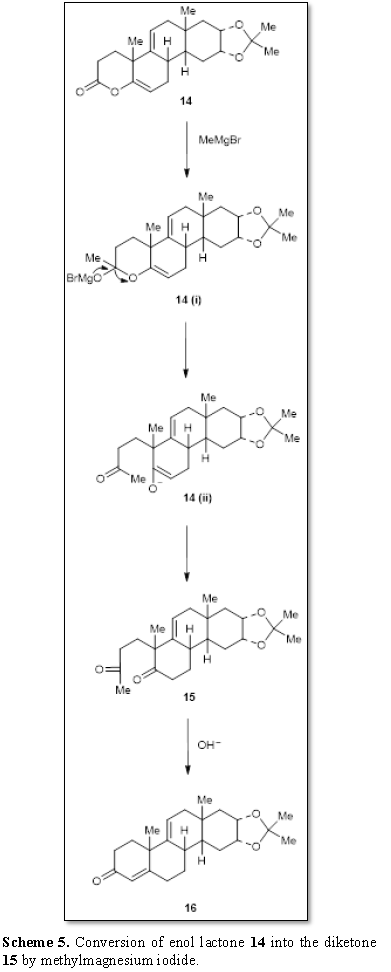
Woodward et al. [10] observed that the enol
lactone 14 can be easily converted
to the diketone 15 by
methylmagnesium iodide (Scheme 5). The
intermediate 14(i) is formed by the
reaction of the Grignard reagent with the lactone 14. The ring cleavage occurred yielding the intermediate enolate
anion 14(ii) and finally to ketone 15 whose conversion to α,
β-unsaturated ketone 16 was effected
with base. It can be observed that a new method for the synthesis of an
unsaturated ketone was developed by the reaction of the lactone with the
Grignard reagent.
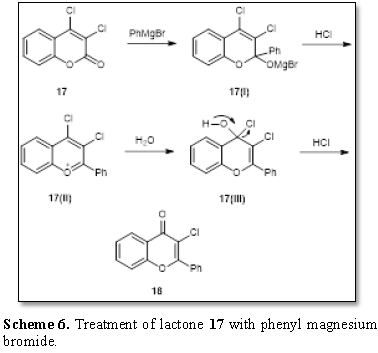
A very different result was obtained when the
lactone 17 was treated with phenyl
magnesium bromide [11]. The product obtained was identified as
3-chloroflavone 18 (Scheme 6). The
mechanism of the transformation is described in Scheme 6. Phenyl magnesium bromide attacks the lactone 17 and resulting intermediate 17(i) with HCl probably forms the
intermediate 17(ii) which is
attacked by water at position 4 to
form the intermediate 17(iii) and
finally the chloroflavone 18.
With epoxide
The epoxides can easily be cleaved by the
Grignard reagents to yield alcohols. The known tendency for oxiranes to undergo
rearrangement during Grignard reactions has been recorded [12]. Many steroidal
epoxides have been cleaved by Grignard reagents to obtain alcohols. It is
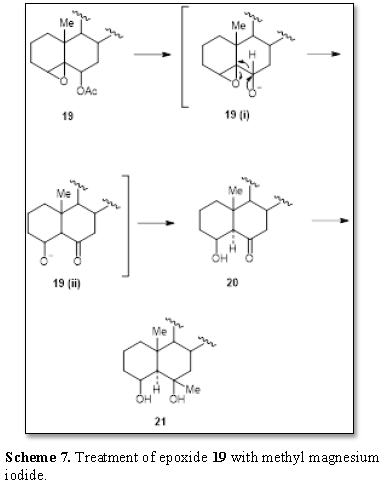
worthwhile to cite the work of Bull [13]. The epoxide 19 with methyl magnesium iodide afforded the β-alcohol 21 (Scheme 7). It can be observed that
the proton attached to the acetate-bearing carbon atom cleaved the epoxide
yielding the intermediates 19(i) and
19(ii) and finally afforded the
β-ketol 20 which then underwent
alkylation by the Grignard reagent to produce the diol 21.
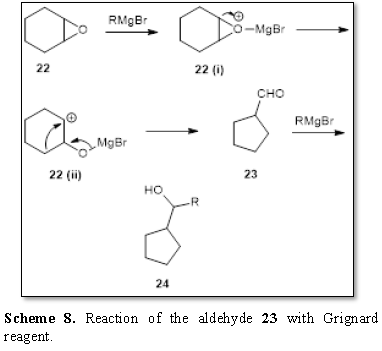
It has been observed that the some epoxides
undergo cleavage along with contraction of rings [14]. Thus the epoxide 22 on treatment with Grignard reagent
yielded the aldehyde 23 through the
intermediate 22(i) and 22(ii). Further the reaction of the
aldehyde 23 with Grignard reagent
yielded the alcohol 24 (Scheme 8).
The above mentioned examples convinced us
that the formation of the final product by the Grignard reagent depends on the
presence of functional group present in organic molecule. If the organic
molecule contains more than one functional group then it is very difficult to
expect normal product. Many examples can be cited which exhibit the formation
of many unexpected products during the reaction of the Grignard reagent with
organic compounds. In this short review due to the space limitation it is not
possible to illustrate many examples.
ACKNOWLEDGEMENT
Dedicated with
affections to the memory of Prof. Anselme JP, editor of the Journal, Organic
Preparations and Procedures Internationals.
The authors gratefully acknowledge the
collaborations of Mr. José Gregorio and Mr. Luis Mujica for literature
references.
CONFLICTS OF
INTEREST
The authors declare that there are not
conflicts of interest.
1. Paquette
LA (2000) Encyclopedia of reagents for organic synthesis. John Wiley &
Sons, West Sussex PO1985Q: England pp: 3532-3535.
2. Ahluwalia
VK, Parashar RK (2003) Organic reaction mechanisms. Alpha Science International
Ltd.: UK, pp: 461-509.
3. Julia
M, Julia S, Guegan R (1959) On a recurrence method for the reformation of
isoprenic chains. Comp Rend 248: 820-822
4. Roberts
JD, Mazur RH (1951) Interconversion reactions of cyclobutyl, cyclopropyl
carbonyl and allylic carbonyl derivatives. J Am Chem Soc 73: 2509-2520.
5. Tsuruta
H, Mukai T (1968) The Grignard reaction of 3-halotropolone methyl ethers. Bull
Chem Soc Jpn 41: 2489-2494.
6. Forbes
EJ, Warrell DC, Fry WJ (1967) The action of alkali on 2-halogenotropanes. J
Chem Soc (C) 1967: 1693-1696.
7. Fusion
RC, Friedlander WS (1953) Displacement of substituents in phenyl 2, 4,
6-triisopropylphenl ketone by the action of Grignard reagent. J Am Chem Soc 75:
5410-5411.
8. Kanjilal
PR, Alam SK, Ghatak UR (1981) An improved total synthesis of (±)-podocarpic
acid by a cycloalkylation route. Synth Commun 11: 795-802.
9. Haworth
RD, Moore BP (1946) Synthesis of 6- and
7-methoxy-1:12-dimethyloctahydrophenanthrene-1-carboxylic acids J Chem Soc
633-636.
10. Woodward
RB, Sondheimer F, Taub D, Heusler K, McLamore WM (1952) The total synthesis of
steroids. J Am Chem Soc 74: 4223-4251.
11. Newman
MS, Ferrai JL (1962) One-step conversion of 3, 4-dichlorocoumarins to
3-chloroflavones. Tetrahedron Lett 3: 199-201.
12. Christensen
BG, Strachen RG, Trenner NR, Arison BH, Hirschmann R, et al. (1960) The partial
synthesis of 12α-methyl-11-dehydrocortic-osterone. J Am Chem Soc 82: 3995-4000.
13. Bull
JR (1969) Anomalous Grignard reactions of steroidal epoxides. J Chem Soc 1969:
1128-1134.
14. Rickborn
B, Gerkin RM (1971) Lithium salt catalyzed epoxide-carbonyl rearrangement
alkyl-substituted epoxides. J Am Chem Soc 93: 1693-1700.